Introduction
Human adaptation and other manifestations of Natural selection have helped researchers to unravel various biological mechanisms of defiance to infectious diseases. Pathogens are infections or infectious agents that cause fallibility and mortality among mankind and other animals globally. While migrating all over the world, the human population have come across some definite pathogens and natural selection has increased the pervasiveness of the resistance alleles to evolve and spread over time. With the help of modern edge analytical methods, advanced technology and extensive resources of public data, the natural selection is unleashing some unique intuition regarding the functioning mechanism of human biology. In this present study we will have an overall idea about how natural selection has affected the evolution of Pathogen Genomes and what measures have been taken to resolve the infectious diseases in UK through years.
Natural Selection Models
Natural selection is the process by which propitious genetic traits increase in abundance over time, whereas the hostile genetic traits lose their abundance (Bentley & Parkhill, 2015). To address this process several different models have been proposed for different values of parameters concerned, like- mode of inheritance, tenacity of selection etc. (Biek et al., 2015) All of the selection models possess two loci having two alleles and the selection takes place on the basis of fitness among the 3 genotypes (one heterozygote and two homozygotes).
- Directional Selection Model- until getting stability one of the two alleles is preferred over the other and the abundance is also increased through time (Croll et al., 2015).
- Balancing Selection Model- in this method a new allele has Quick and Stable equilibrium frequency. Compared to both the homozygotes, the heterozygote has a greater fitness (Delong et al., 2016).
- Diversifying Selection Model- in this classical model, two or more phenotypes are preferred simultaneously giving rise to an increased pattern in genetic variation (Croll et al., 2015).
Tests to identify Natural Selection
Selection has different impacts on genetic variations. In a simple Directional Selection model, novel mutation will be preferred if it is concerned about some selective advantages of the pathogen genome (Dong, Raffaele & Kamoun, 2015). This phenomenon is termed as Positive Selection. Negative selection occurs if the mutation seems dangerous to the genome. Both the selection processes can be measured on a single amino acid of the gene or across the whole genome. 3 major procedures will be discussed further to infer selection methods in pathogen genomes (Myers & Cory, 2016).
- Substitution rate patterns- genes in microbial genomes generally mutate independently with different populations. In response to common Selection Pressure, a convergent or parallel evolution is identified by intermittent emergence of same substitution within different individuals (Porse et al., 2016). During Tuberculosis treatment, by antimicrobial drugs selective pressure is applied on the pathogen Mycobacterium tuberculosis which is evident from the fact that the same drug resistance point mutation within different bodies emerges frequently. The number of substitution per gene can be easily calculated by multiplying the gene’s length and mutation per site (Pugliese et al., 2016). Thus, any significant rise in the substitution rate than from the expected value can be used as a clear indication of positive selection.
- dn/ds estimation- another popular method for the detection of selection between divergent individuals is dn/ds estimation. dn stands for the rate of non-synonymous substitution per non-synonymous site, where as ds is the counterpart for the synonymous ones (Quainoo et al., 2017). In the absence of selection dn/ds ratio remains zero, i.e, it signifies a neutral condition. If the value exceeds 1, it indicates a positive selection and when the value is less than 1, it suggests a negative selection (Sánchez et al., 2018). However, there are some restrictions while applying this model for microbial populations (Croll et al., 2015). Firstly, if the expected number of substitution at any individual is very small, then it will be difficult calculate the non-neutral dn/ds. Secondly, due to higher existence of negative selection sites, the true value of dn/ds goes below the expected value which reduces the statistical power of positive selection site detection (Stukenbrock, 2016).
- MacDonald Krietman Test- the MK test is the assessment for the ratio of Pn/Ps and Dn/Ds. Pn/Ps is the ratio of non-synonymous to synonymous polymorphisms. Dn/Ds is the ratio of non-synonymous to synonymous fixed contrasts (Stukenbrock, 2016). As per the comment of Quainoo et al., (2017), this particular test is widely used to test within species selection type but it can also be used to decipher selection among the sub-populations. Dn/Ds> Pn/Ps indicates that the non-synonymous changes are higher than the fixed differences, suggesting a positive selective, while negative selection is inferred when Dn/Ds< Pn/Ps (Croll et al., 2015).
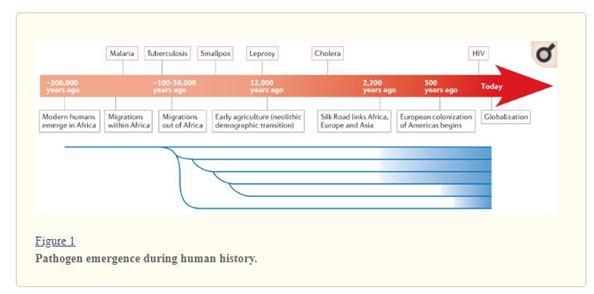
Figure: Evolution of Human Pathogenic genomes
(Source: Quainoo et al., 2017)
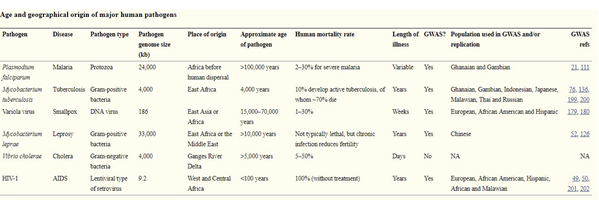
Figure: origin of human pathogen
(Source: Pugliese et al., 2016)
From the age of modern humans, there has been a trend of increase in human pathogens as well (Porse et al., 2016). Some pathogens are responsible for large human casualties, like- the bacteria Yersinia pestis causing Black Plague, the virus Variola responsible for Smallpox, the Malaria protozoa Plasmodium etc. Just after the migration from Africa human lineage broke into fragments both geographically as well as genetically (Dong, Raffaele & Kamoun, 2015). Gradually, theses population started to mix through colonization and the population increased. On closer contact with animals and people all over the world, infectious pathogens became widespread. As a consequence, fatal diseases like, Malaria, Cholera, Smallpox, Tuberculosis, Leprosy became more common. As the civilization is developing day by day, mankind is facing more severe threats like HIV, AIDS, SARS (Myers & Cory, 2016).
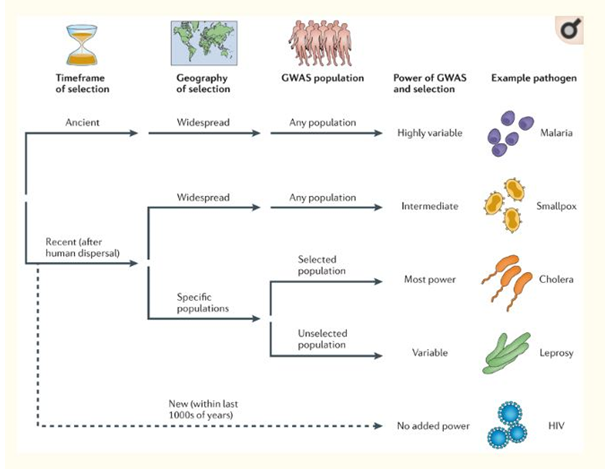
Figure: GWAS population
(Source: Dong, Raffaele & Kamoun, 2015)
Pathogen genomics: measures and initiatives taken by UK
The study of pathogen genomics has been globally given importance for better understanding, management and investigation of infectious diseases. The main pathogenic viruses in the UK are Human Immunodeficiency Virus or HIV and Hepatitis C virus (HCV) (Dong, Raffaele & Kamoun, 2015). HIV is responsible for the slow, progressive and most fatal disease that is Acquired Immunodeficiency Syndrome or AIDS. Not only in UK, but AIDS has become the most alarming global health burden nowadays, resulting almost a million deaths worldwide within a year. Statistics reveal that annually around 500 people die due to AIDS in UK. Medical survey states that in 2012, almost a million people of UK were infected with HIV and among them 20-22% were not even aware of that fact (Dong, Raffaele & Kamoun, 2015).
Whole Genome Sequencing techniques have been discovered to resolve the pathogen related diseases. For this purpose, several initiatives have been taken and large number of publications has been published. Chief Medical Officer’s Annual Report, 2016 is one of them (Dong, Raffaele & Kamoun, 2015). European countries have taken some serious measures on this particular issue. The Public Health England (PHE) has been trying to implement modern WGS techniques in order to secure a healthy future. The National Infection Service (NIS) of UK has a national strategy of WGS to intensify the control of pathogenic diseases and to help PHE reach its goals in infectious disease management (Dong, Raffaele & Kamoun, 2015). At PHE, “Colindale” a centralised WGS service has been established to provide services for a range of nationally important pathogens including E. coli, Shigella, Listeria, Campylobacter, S. aureus, Salmonella and Mycobacteria. The Colindale WGS service of UK was one of the prime UK laboratories to get appraisal for its Whole Genome Sequencing services of virus and bacteria. Among the initial adopters of the Whole Genome Sequencing approach for conventional services delivery at PHE was the GBRU- Gastrointestinal Bacteria Reference Unit. It was the National Reference laboratory for Gastrointestinal bacterial pathogens. Gastrointestinal (GI) disease is the illnesses mainly caused by foodborne transmission of bacteria or via being in direct touch with animals and their environments (Delong et al., 2016). UCL has another pathogenic unit established by the grants from UK’s Medical Research Council (MRC). The unit carries out the following:
- Library preparation for whole genome sequencing (Delong et al., 2016)
- Library preparation for pathogen genome enrichment including clinical samples
- RNA sequencing for pathogen discovery
- Amplicon sequencing (Delong et al., 2016)
- Viral resistance gene amplification
- Bioinformatics
- Sequence analysis (Delong et al., 2016)
Conclusion
As
pathogens’ genetic sequence changes over time, they are generally small and
have a rapid evolving rate; this is the only reason that they are effective and
persistent agents of causing infectious diseases. Natural selection plays a
major role in investigating the evolution of such pathogenic genomes.
Scientists have discovered that ancient characteristics of balancing selection
along with the most recent characteristics of positive selection equally
incriminate the pathogens as one of the strongest selective causes of human
evolution. Both the birth and death rates are thus dependent on the evolution
pathogenic genomes. UK has initiated some good measures to deal with such
infectious diseases carried by pathogenic genomes and has also taken
initiatives to spread awareness about the issue globally.
Reference List
Bentley, S. D., & Parkhill, J. (2015). Genomic perspectives on the evolution and spread of bacterial pathogens. Proceedings of the Royal Society B: Biological Sciences, 282(1821), 20150488.
Biek, R., Pybus, O. G., Lloyd-Smith, J. O., & Didelot, X. (2015). Measurably evolving pathogens in the genomic era. Trends in ecology & evolution, 30(6), 306-313.
Croll, D., Lendenmann, M. H., Stewart, E., & McDonald, B. A. (2015). The impact of recombination hotspots on genome evolution of a fungal plant pathogen. Genetics, 201(3), 1213-1228.
Delong, T., Wiles, T. A., Baker, R. L., Bradley, B., Barbour, G., Reisdorph, R., … & Elso, C. M. (2016). Pathogenic CD4 T cells in type 1 diabetes recognize epitopes formed by peptide fusion. Science, 351(6274), 711-714.
Dong, S., Raffaele, S., & Kamoun, S. (2015). The two-speed genomes of filamentous pathogens: waltz with plants. Current opinion in genetics & development, 35, 57-65.
Myers, J. H., & Cory, J. S. (2016). Ecology and evolution of pathogens in natural populations of Lepidoptera. Evolutionary applications, 9(1), 231-247.
Porse, A., Schønning, K., Munck, C., & Sommer, M. O. (2016). Survival and evolution of a large multidrug resistance plasmid in new clinical bacterial hosts. Molecular biology and evolution, 33(11), 2860-2873.
Pugliese, M., Loreto, V., Pompei, S., & Tria, F. (2016). Exploring the evolution of pathogens organised in discrete antigenic clusters. Journal of Statistical Mechanics: Theory and Experiment, 2016(9), 093306.
Quainoo, S., Coolen, J. P., van Hijum, S. A., Huynen, M. A., Melchers, W. J., van Schaik, W., & Wertheim, H. F. (2017). Whole-genome sequencing of bacterial pathogens: the future of nosocomial outbreak analysis. Clinical microbiology reviews, 30(4), 1015-1063.
Sánchez-Vallet, A., Fouché, S., Fudal, I., Hartmann, F. E., Soyer, J. L., Tellier, A., & Croll, D. (2018). The genome biology of effector gene evolution in filamentous plant pathogens. Annual review of phytopathology, 56, 21-40.
Stukenbrock, E. H. (2016). The role of hybridization in the evolution and emergence of new fungal plant pathogens. Phytopathology, 106(2), 104-112.
Stukenbrock, E. H. (2016). The role of hybridization in the evolution and emergence of new fungal plant pathogens. Phytopathology, 106(2), 104-112.